Listen to the conference call to learn more about the merger.
Pipeline of Small Molecule Therapeutics
InMed Pharmaceuticals (NASDAQ: INM) is a clinical-stage pharmaceutical company focused on developing a pipeline of proprietary small molecule drug candidates targeting the CB1/CB2 receptors. InMed’s pipeline consists of three separate programs in the treatment of Alzheimer’s, ocular and dermatological indications.

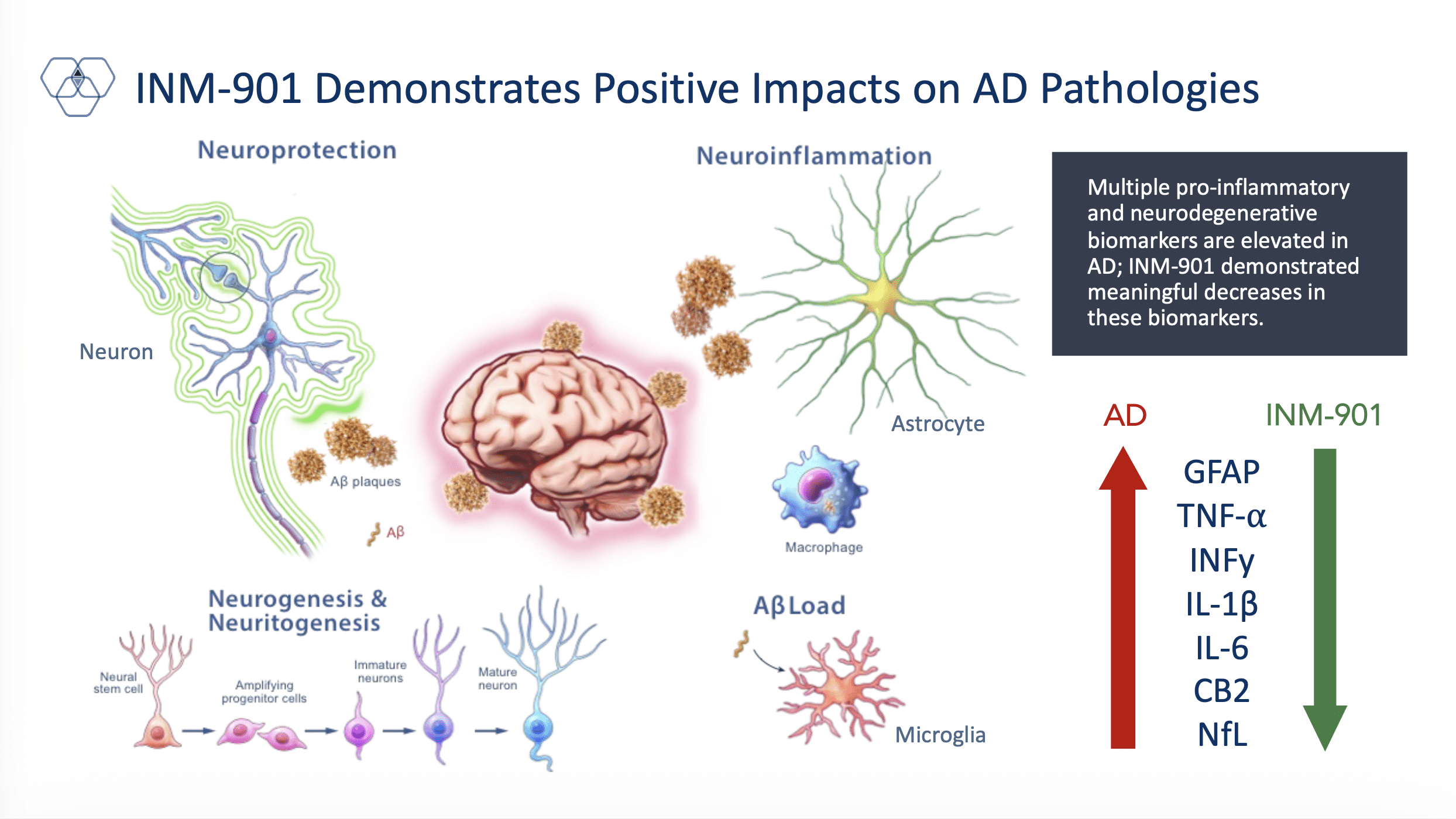
INM-901 targets the reduction of neuroinflammation associated with Alzheimer’s disease
INM-901 is a proprietary, orally bioavailable, disease-modifying small molecule drug candidate that is a preferential CB1/CB2 signaling agonist and can cross the blood-brain barrier with a specific focus on treating neuroinflammation in Alzheimer’s disease. InMed believes INM-901 is uniquely positioned within the evolving Alzheimer’s disease treatment landscape as increasing scientific consensus suggests that the disease is driven by multiple, interrelated biological pathways, rather than a single pathogenic mechanism.
INM-089 for Dry AMD Demonstrates Functional and Pathological Improvements in Preclinical Study
INM-089 has demonstrated its ability to proactively protect the nerve cells in the back of the eye in the retinal area, exhibiting its therapeutic potential in dry age-related macular degeneration (AMD).
Preclinical studies of INM-089 demonstrated significant functional and pathological improvements in a dry AMD disease study model. Results from studies demonstrate that INM-089 provides neuroprotection of photoreceptors, improves photoreceptor function and the integrity of the retinal pigment epithelium and reduces extracellular auto fluorescent deposits, a hallmark of dry AMD.
Experienced team with a successful track record
Together, InMed and BayMedica bring extensive know-how in pharmaceutical research and development and manufacturing. We’ve combined our complementary expertise to become a leading pharmaceutical R&D company and manufacturer of small molecule therapeutics targeting CB1/CB2 receptors.



